 
Its chemical symbol is As, and it was first clearly. Because the two words have defined different sets of elements over the years, the IUPAC recommends the numerical system. The term 'family' is synonymous with the term 'group'. Atoms or ions with the same electronic configurations are said to be. Isoelectronic chemical species typically display similar chemical properties. The term means 'equal electric' or 'equal charge'. The molar mass is calculated in two ways. Isoelectronic refers to two atoms, ions, or molecules that have the same electronic structure and the same number of valence electrons. Avogadro's number tells the number of particles present per mole. This article considers the origin of the elements and their abundances throughout the universe. Elements are the fundamental materials of which all matter is composed. Chemical families tend to be associated with the vertical columns on the periodic table. The periodic table provides the atomic masses used in the calculation of the molar mass. chemical element, any substance that cannot be decomposed into simpler substances by ordinary chemical processes. Arsenic, a chemical element in the nitrogen group (Group 15 Va of the periodic table), existing in both gray and yellow crystalline forms. In chemistry, a family is a group of elements with similar chemical properties. The modern understanding of organic and inorganic chemistry is not consistent with these historical meanings. Several complex organic compounds of arsenic have been employed in the treatment of certain diseases, such as amebic dysentery, caused by microorganisms. But, this definition is not completely correct because the field of Inorganic Chemistry also. An illustration of the historic meaning of "organic" and "inorganic". The names 'organic' and 'inorganic' come from science history, and still today a generally-accepted definition of Inorganic Chemistry is the study of non-carbon molecules, or all the elements on the periodic table except carbon ( Figure 1.1.1 1.1. Unfortunately, there was a slightly different system in place in Europe.\). The first two groups are 1A and 2A, while the last six groups are 3A through 8A. 
The traditional system used in the United States involves the use of the letters A and B. There are two different numbering systems that are commonly used to designate groups, and you should be familiar with both. In a group, the chemical elements have atoms with identical valence electron counts and identical valence vacancy counts. The most reactive elements and compounds may ignite spontaneously or explosively. The reaction can involve the substance on its own or with other atoms or compounds, generally accompanied by a release of energy. These two rows are pulled out in order to make the table itself fit more easily onto a single page.Ī group is a vertical column of the periodic table, based on the organization of the outer shell electrons. Group, in chemistry, a column in the periodic table of the chemical elements. In chemistry, reactivity is a measure of how readily a substance undergoes a chemical reaction. Periods 6 and 7 have 32 elements, because the two bottom rows that are separate from the rest of the table belong to those periods. 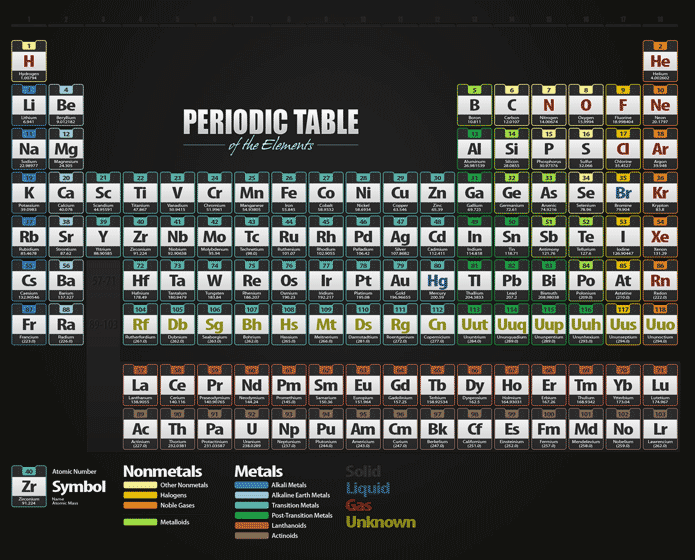
Period 1 has only two elements (hydrogen and helium), while periods 2 and 3 have 8 elements. An example of an element family is the nitrogen group or pnictogens. Learn more about noble gases with this article. All of the noble gases are present in Earth’s atmosphere and are colorless, odorless, tasteless, and nonflammable. Most element families are a single column of the periodic table, although the transition elements consist of several columns, plus the elements located below the main body of the table. The seven elementshelium, neon, argon, krypton, xenon, radon, and oganessonof Group 18 of the periodic table. A new period begins when a new principal energy level begins filling with electrons. Element families are elements that have the same number of valence electrons. There are seven periods in the periodic table, with each one beginning at the far left. \) (Credit: User:Cepheus/Wikimedia Commons Source: (opens in new window) License: Public Domain)Ī period is a horizontal row of the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
